Projects
Identification of protein networks associated with GABA receptors
Over the recent years it became evident that G-protein coupled receptors as well as ionotropic receptors and ion channels are clustered in supra-molecular complexes via networks of protein-protein interactions. These multi-protein networks are implicated in receptor targeting to specific cellular compartments, in their trafficking and in the assembly and fine-tuning of the signalling machinery. We think that GABA receptors are organized in large protein complexes or protein networks that are required for signal transduction. Given the heterogeneity of GABA receptors there may exist in brain a variety of distinct protein networks associated with GABA receptors. We consider these “GABA receptor signaling complexes” as dynamic structures, which may alter their constituent proteins in different physiological states or under pathophysiological conditions. However, information on such “GABA receptor signalling complexes” is largely lacking. Therefore, we follow up two approaches to identify proteins associated with GABA receptors:
- First, we screen for GABA receptor interacting proteins using the yeast two-hybrid system. This is a powerful and very sensitive technique to identify direct protein-protein interactions but it is, however, not very suitable for analyzing multi-protein networks.
- Second, we are working on the implementation of proteomic approaches combining affinity chromatography and/or co-immunoprecipitation with mass spectrometry (MS) to directly identify and characterize multi-protein complexes associated with GABA receptors.
Trafficking of GABA(B) receptors
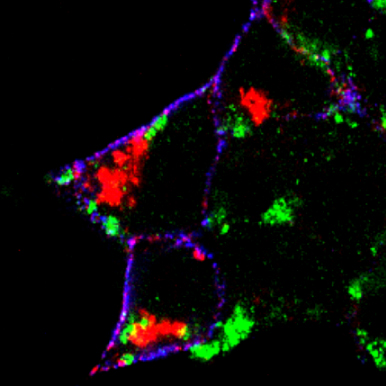
Receptor internalization is now recognized as an important mechanism for rapidly regulating cell surface numbers of receptors. However, there are conflicting results on the existence of rapid endocytosis of GABA(B) receptors. To gain insights into trafficking of GABA(B) receptors we established assays based on immunocytochemical, cell surface ELISA and biotinylation methods for analyzing GABA(B) receptor trafficking qualitatively and quantitatively. In addition, we are interested in live cell imaging to analyze real time trafficking of GABA receptor. As experimental systems we are using HEK 293 cells transiently expressing the proteins of interest as well as primary cultured neurons.
Techniques available in the lab
- Basic biochemical and molecular cloning methods
- Production of polyclonal antibodies
- Pharmacological characterization of receptors by radioligand binding studies
- Receptor autoradiography
- Cell culture / cell transfection
- Western blotting / immunoprecipitation
- Two-dimensional polyacrylamide gel electrophoresis
- Protein purification techniques
- Over expression of proteins
- Yeast Two Hybrid screenings
- Single and multiple immunofluorescence staining of cell cultures
- Confocal laser scanning microscopy