Oligodendrocytes in Painful Diabetic Neuropathy
Introduction
Our efforts focus on unraveling the effect of diabetes on myelin homeostasis and its implication on the pathophysiology of painful diabetic neuropathy.

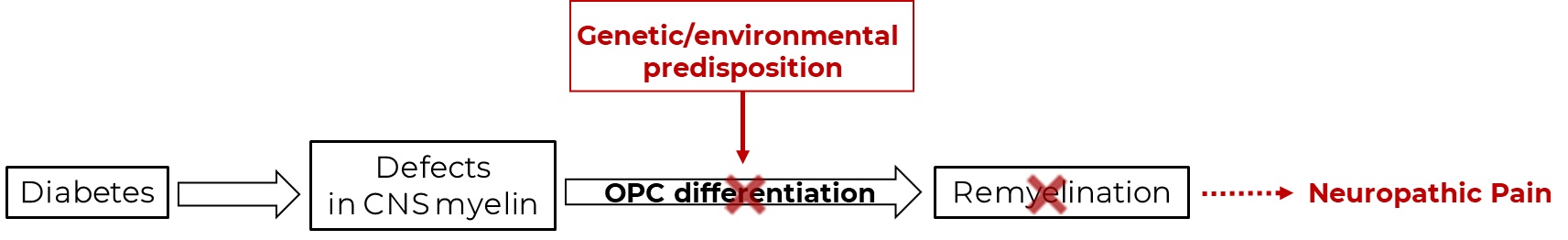
Diabetes mellitus is a pandemic that has expanded enormously over the past 2 decades. Peripheral diabetic neuropathy is a common, serious, chronic complication of the disease affecting up to 50% of the patients. Among them, 40-50% further develop painful diabetic neuropathy. It is not clear which factors predispose only a subset of patients to the development of neuropathic pain, but clinical studies have identified several risk factors, including female gender, obesity, high alcohol consumption, lower vitamin D levels, and advanced age.
All these factors suppress oligodendrocyte precursor cell (OPC) differentiation and subsequent myelin formation. Furthermore, several of the genes associated with the development of diabetic neuropathy are expressed in oligodendrocytes, with some of them implicated in OPC maturation.
Although peripheral demyelination stands among the well-described processes associated with the pathogenesis of diabetic neuropathy, accumulating evidence suggests that diabetes also affects myelin dynamics in the central nervous system. On the other hand, depletion of mature myelinating oligodendrocytes, per se, triggers pain-like behavior in mice. Interestingly, one of the most widely used drugs for the treatment of type 2 DM, metformin, except from its protective effects on peripheral nerve degeneration, has been shown to promote OPC differentiation and appears to have antinociceptive effects in several rodent pain models.
Considering collectively the above-stated evidence, our main hypothesis is that diabetes causes central demyelination. Consequently, new myelin formation and OPC differentiation/maturation are required for repair. Factors that impede this process predispose a subset of patients to the development of central pain.