Navigation auf uzh.ch
Navigation auf uzh.ch
Our previous studies in healthy adults revealed that the distinct EEG effects of physiologically elevated sleep need following sleep deprivation and allosteric agonistic modulation of GABAA receptors are mediated by different neurophysiological mechanisms. In contrast, our work on the serotonergic system showed that altering the balance between 5-HT2 and 5-HT1A receptor activity may be necessary, but is not sufficient, for inducing the physiological changes in the sleep EEG, which are typically seen after sleep deprivation.
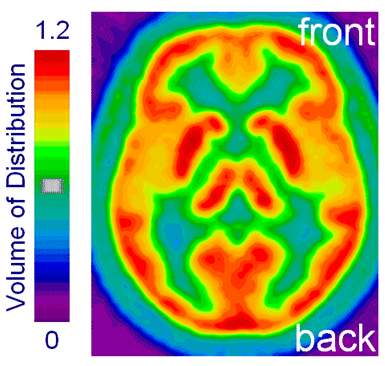
A more direct relationship between sleep homeostasis and neurochemical changes in the brain may be present in the adenosinergic system. We demonstrated that the adenosine A1 and A2A receptor antagonist, caffeine, reduces EEG markers of elevated sleep need in wakefulness and sleep. Moreover, we provided the first direct evidence that the adenosinergic system modulates sleep and EEG correlates of sleep homeostasis in humans. We observed that a functional genetic variation in the adenosine metabolizing enzyme, adenosine deaminase, affects duration and intensity of deep slow wave sleep. More recent results suggest that adenosinergic mechanisms modulate individual differences in waking-induced changes in neurobehavioral functions and brain rhythmic activity. These findings further support an important role of adenosine and adenosine receptors in sleep-wake regulation. Consistent with this notion, we found high A1 receptor density in cortical and subcortical structures known to be involved in regulating sleep-wake functions (Fig. 1).
Another line of research revealed that genetic variation in the adenosine A2A receptor gene (ADORA2A) contributes to individual sensitivity to subjective and objective effects of caffeine on sleep. Moreover, a distinct ADORA2A haplotype predicts fast performance on a sustained vigilant attention task, which reliably reflects elevated sleep need.
Taken together, these data indicate that the adenosinergic system may be a possible target for the pharmacological improvement of disturbances of sleep and vigilance, which are highly prevalent in the general population.
The dissipation of low-frequency EEG activity reflecting sleep need flattens with increasing age. Furthermore, investigation of local EEG changes showed that aging not only reduces global EEG power during sleep, but also causes frequency-specific alterations in sleep EEG topography. These results support the concept of sleep as a local process. Ongoing projects investigating age-related changes in waking and sleep demonstrate prominent alterations in healthy men in the vulnerability of neurobehavioral functions to impairment by sleep loss. These findings have important implications for the prevention of sleep-related accidents, which are recognized today as a major public health issue associated with immense economic costs for society (Fig. 2).
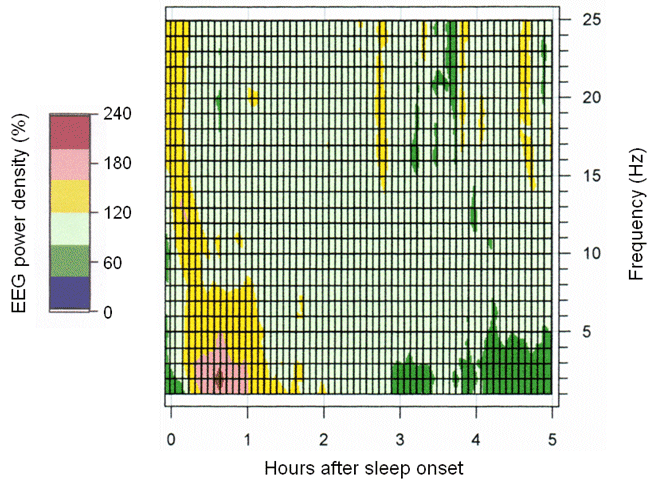
To test the hypothesis that sleep regulation is altered in major depression, we studied topographic changes in the EEG during sleep. Our findings did not support impaired sleep regulation in moderately depressed patients. We showed that REM sleep can be pharma¬cologically eliminated with an effective antidepressant without altering the physiological decline of low-frequency activity in non-rapid-eye-movement (nonREM) sleep (Fig. 3). These results provided clear evidence that nonREM sleep and rapid-eye-movement (REM) sleep are regulated by separate processes.
Further studies showed that the expression of REM sleep is controlled by serotonergic mechanisms. We developed the first non-disruptive, double-blind method for studying human subjects overnight with and without REM sleep. This method opens-up a novel strategy for investigating the functions of REM sleep, and the roles of serotonin and REM sleep in the regulation of, for example, nonREM sleep, memory and mood.
To unravel other mysteries of sleep and possible pathophysiological mechanisms underlying disturbed sleep-wake functions, we are currently studying patients with idiopathic hypersomnia, narcolepsy and sporadic Creutzfeldt-Jakob Disease in an interdisciplinary network of neuroscientists and clinicians.