Navigation auf uzh.ch
Navigation auf uzh.ch
We are interested in understanding how glial cells and neurons interact to maintain normal brain function and long-term cellular homeostasis. The mechanisms by which glial cells are involved in regulating neuronal functions and metabolism are still poorly understood. Current working models suggest that both myelinating oligodendrocytes and astrocytes, which form a pan-glial syncytium by gap-junction coupling, are highly glycolytic and supply neuronal compartments with energy substrates to fuel energy demands and to support long-term integrity. We are particularly interested in how myelinated axons are maintained by glial support functions. Very little is known about the cellular mechanisms underlying axon-glial interactions and metabolic cooperation. How do myelinating oligodendrocytes sense neuronal activity and how are these signals translated into maintaining neuronal functions? How are these cellular interactions perturbed during inflammation, normal aging and upon neurological pathologies? And could perturbations in axon-glial communication and deficits in glial metabolic support impact the etiology and pathogenesis of neurodegenerative diseases? To address these questions we combine molecular genetics, electrophysiology, in vivo and ex vivo two-photon imaging, histology, electron microscopy and behavioral studies in mice to investigate cellular mechanisms regulating intercellular communication, energy homeostasis and cellular integrity.
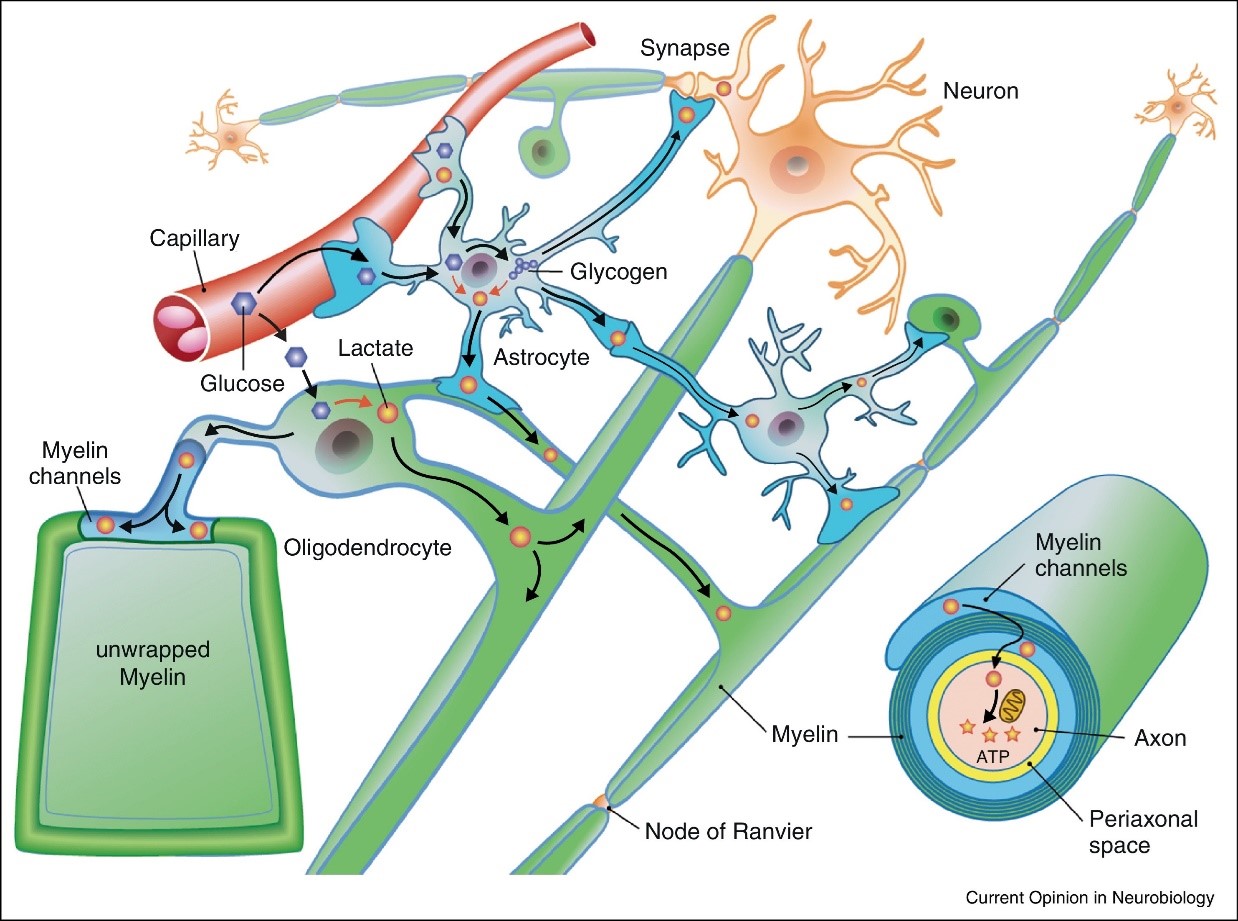
Scheme of neuron-glia interactions and metabolic support to neuronal compartments. Myelinated axons may receive lactate from glycolytic oligodendrocytes through myelin channels, whereas synapses and dendrites are supported by astrocytes through their contacting processes. Oligodendrocytes and astrocytes are gap junction-coupled forming a “pan-glial syncytium” that is connected to brain capillaries which may also serve as a route for metabolites to axons and dendrites. Figure taken from Saab et al., 2013. https://doi.org/10.1016/j.conb.2013.09.008